REJOIN Medical
Chirurgische Instrumente – neu interpretiert.

Fallstudie über die intensive Design-Kooperation mit Rejoin / Mastin Medical, einem neu gegründeten chinesischen Sportmedizin-Start-up, das sich auf arthroskopische Instrumente für die Chirurgie spezialisiert hat.
Innerhalb von drei Jahren erarbeitete unser Shanghai Team gemeinsam mit dem Chinesischen Start-up Unternehmen Mastin/ REJOIN einen komplett neuen und mutigen Auftritt im Markt der Chirurgischen Einmalinstrumente für die Sportmedizin.
Die kantige Formensprache im magentafarbenen, transluzenten Design ist in erster Linie Ergebnis der ergonomischen Untersuchungen und der fertigungstechnischen Optimierung im Hinblick auf die Spritzgusstechnik. So lässt sich eine optimale Griffigkeit erzeugen, die gleichzeitig zu leichten Instrumenten führt, Materialeinsparung und damit Kostensenkung inklusive.
Aufgabe
Vorgehen
Ergebnis

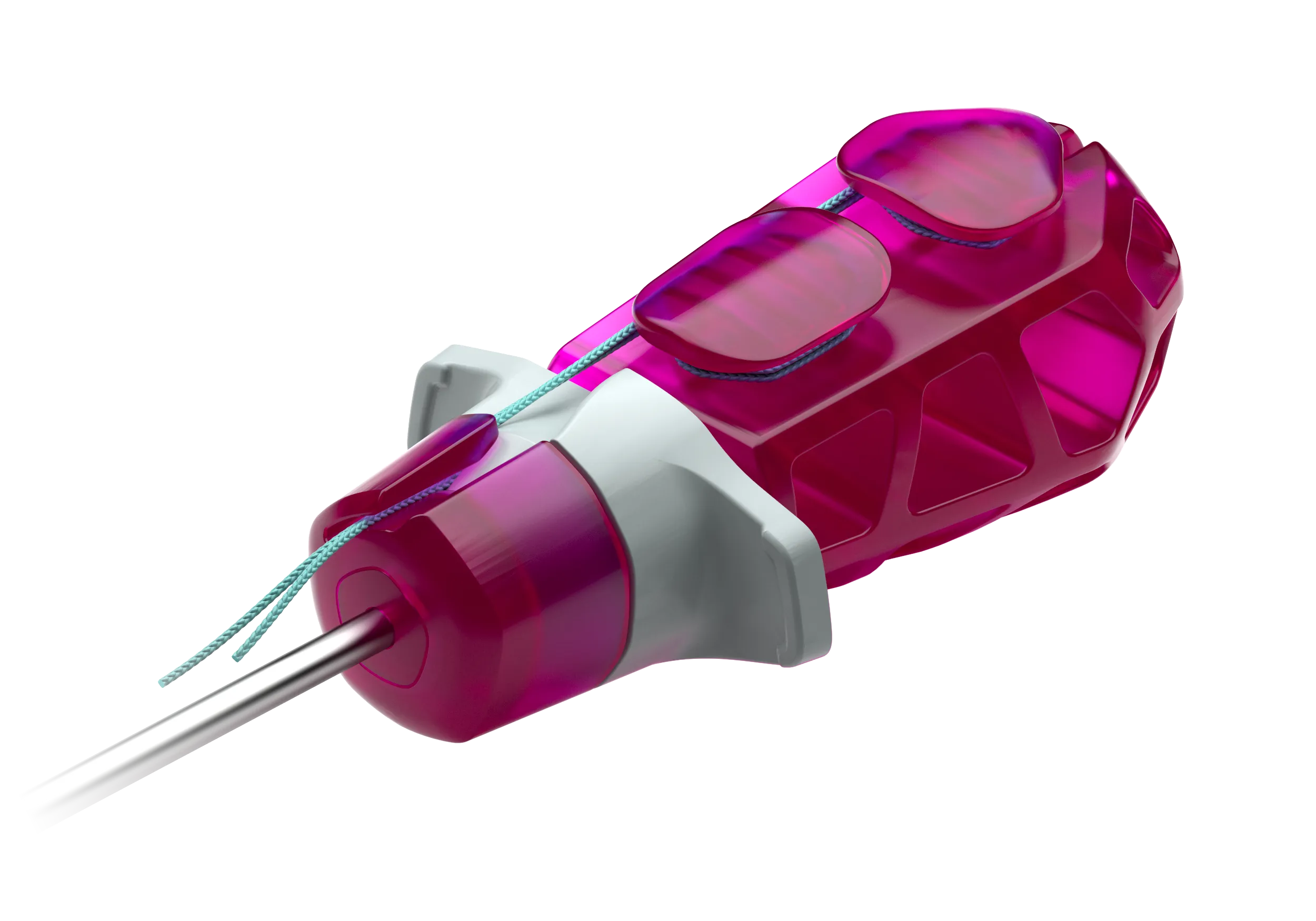
Eine globale sportmedizinische Marke entwickeln
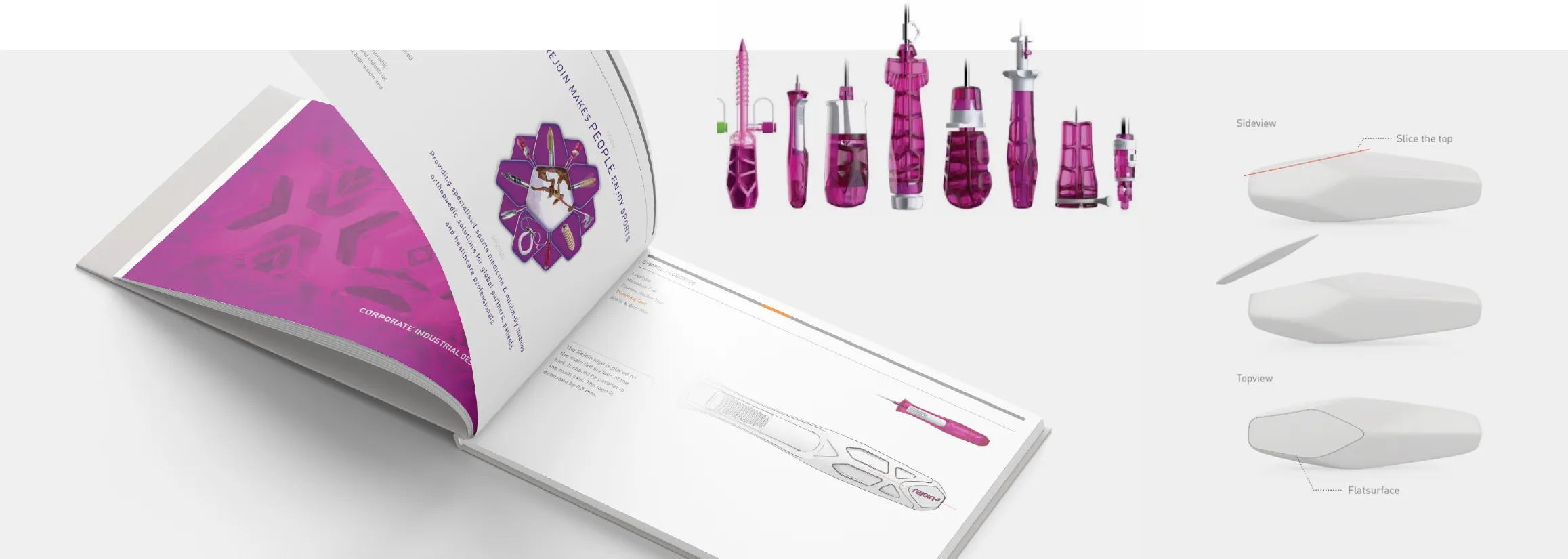
Um eine hohe Wertanmutung mit der Wiedererkennbarkeit der Marke REJOIN zu koppeln, erstellten wir parallel zur Produktentwicklung eine Design Guideline (genannt CIDM - Corporate Industrial Design Manual), die die Übertragung der Designmerkmale auf neue Instrumenten-Typen leicht macht und so eine hohe Einheitlichkeit im Markenerscheinungsbild sicherstellt.
Rejoin benötigte zunächst einmal starke Leitlinien die ihnen genügend Sichtbarkeit verschafften, um sich in einem zunehmend wettbewerbsintensiven Markt zu etablieren. Um mehr über die Ausgangssituation zu erfahren, lesen Sie Teil 1 von „Die Zukunft ist nicht-invasiv", einem exklusiven 3-teiligen Artikel, der ausführlich die Hintergründe beschreibt und warum Rejoin zur richtigen Zeit am richtigen Ort war, um von einem boomenden Sportmedizinmarkt in China zu profitieren.

Priorität #1: Klare Markenstrategie
Von Beginn an mit einem prägnanten Auftritt
Während der ersten Diskussionen über die Rejoin Marke formulierte WILDDESIGN die zukünftige Marktposition von Rejoin als dynamische, sportorientierte medizinische Marke, die sich unter einer Vielzahl von Unternehmen, klinischen oder traditionellen Medizinunternehmen hervorheben sollte. Um diese Annahme mit Rejoins Entwicklerteam zu überprüfen, erstellten wir so genannte Affinity Maps, um die zukünftige Position von Rejoin gegenüber lokalen und globalen Wettbewerbern zu visualisieren. Abgeleitet von diesem ersten Schnappschuss war schnell klar, dass Farbe und Form unsere Treiber für das Design sein würden.
Designsprache
Nach Rejoins okay zu dieser Roadmap war es dann unsere Aufgabe, eine Industrial Design Sprache zu entwickeln und implementieren, aus der einerseits hochfunktionelle und andererseits bestechend ästhetische arthroskopische Werkzeuge für Chirurgen und Krankenhauspersonal abgeleitet werden konnten. Unsere allerwichtigste Aufgabe blieb jedoch die Usability- und Ergonomieanforderungen der Chirurgen und des Krankenhauspersonals zu treffen, besser noch zu übertreffen.
Priorität #2: Gute Usability - das fundamentale Designprinzip
Nach der Teilnahme an diversen User-Workshops, Seminaren und so genannten Produkt-Walk-Throughs mit Chirurgen, um jedes einzelne Instrument wirklich ganzheitlich zu verstehen, konzentrierte sich das WILDDESIGN-Team auf die vorrangigen Usability-Probleme in den Handflächen und Fingerspitzen der Chirurgen.
Mittels Rapid Prototyping in schnellen Iterationen wurden verschiedene Formfaktoren erarbeitet und getestet, die in einem einzigartigen Griffkonzept endeten: ein Griff mit abgerundetem, sich verjüngendem fünfeckigen Querschnitt, der sich in der Hand des Chirurgen ideal anschmiegt und anfühlt. Weiterer 3D-Druck mit zusätzlicher Handmodellierung simulierte die endgültige Oberflächentextur und ermöglichte realistische Tests mit Latexhandschuhen.
Weiteres Prototypen und Testen
Zur weiteren Detaillierung wurden Verrippungs-Versuche angestellt, die dabei halfen, den Rohstoffverbrauch zu reduzieren und gleichzeitig die Taktilität erhöhten. Das schaffte schließlich den Durchbruch zu einer einzigartigen Produktsprache auch wenn es noch ein hartes Stück Arbeit für die Designer war, bis das ganze als ein integrales Teil sauber aus der Spritzgußform entformt werden konnte. Durch Dutzende von Schaum- und 3D-gedruckten Prototypen wurden Ausmaß und Position jedes Durchbruchs definiert, wobei die endgültige Form sowohl den Design- als auch den Konstruktionsanforderungen angepasst wurde. Unsere„Rippenformel“ zur schnellen Ableitung weiterer Griffstücke wurde ebenfalls formuliert und in das Design Guideline Dokument aufgenommen, um sowohl ein komfortables und leichtes Instrument für die Hand des Chirurgen zu gestalten als auch die Materialkosten für den Lieferanten zu reduzieren.

Bedeutung hinter den CMF-Entscheidungen
Angesichts der immensen Flut traditioneller Produkte auf dem Markt, war das Designteam von Anfang an zuversichtlich, Farbe zu einem integralen Bestandteil der Designrichtung machen zu können. Es fehlte nur ein letzter Tick - Transluzenz. Lesen Sie Teil 2 unseres Blogartikels, um einen tieferen Einblick in diesen Teil von WILDDESIGNs Designprozess für Rejoin zu erhalten.
Rejoin-Transluzenz
Mehrfache Lichtbrechungen bringen Tiefe, Sättigung und visuelle Anziehungskraft in das Griffstück, während die sichtbaren inneren Strukturen des montierten Instruments ein Verständnis des inneren Aufbaus schaffen, die wichtige Hinweise zur Einbringung der Implantate und letztendlich zur Druchführung arthroskopischer Verfahren geben können. Aus rein ästhetischen Erwägungen heraus hebt sich ein purpurfarbenes, durchscheinendes Werkzeug, das auf einem Tisch mit einer Vielzahl von anderen opaken Werkzeugen liegt, einfach ab und bildet ein äußerst aufmerksamkeitsstarkes Sinnbild für diesen sportmedizinischen Bereich. In Zusammenarbeit mit spezialisierten Lieferanten von medizinischen Kunststoffen begannen wir dann, die idealen Polymere und Veredelungen auszuwählen, um dieses Konzept in die Produktion zu bringen.

Vorbereitet für die Zertifizierung
Um sicherzustellen, dass unsere Designs für REJOIN reibungslos in den medizinischen Markt gebracht werden können, hat das WILDDESIGN-Team eng mit erstklassigen Kunststofflieferanten und mit dem QM-Team von REJOIN zusammengearbeitet. Da es sich um medizinische Produkte der Klasse III handelt, Produkte mit hohem Risiko, normalerweise für implantierbare oder lebenserhaltende Systeme vorgesehen, ist das Prüfniveau für die Bewertung der Produkte extrem anspruchsvoll.
Medizinische Zulassung
Mit dem Verständnis des Time-to-Market und einer angestrebten Zulassungszeit von 14 Monaten, testete das Entwicklungsteam das Polymer in Spritzgußformen, um den CFDA-Anforderungen zu entsprechen und Garantien bei den Drittanbietern zu erwirken. Letztlich konnte REJOIN seinen Entwicklungsplan einhalten und die geplanten Meilensteine in seiner Produktpipeline alle erreichen. Aus der Führungsebene heraus kam die Vision, die Marke Rejoin zum Marktführer zu machen und dafür eine einzigartige Designsprache als Transportmittel zu verwenden. Wir durften diese Sprache dann auf alle zukünftigen Instrumente übertragen, um die Markenwerte zu stärken und das Vertrauen in die globale Entwicklung zu verankern.

Neue & verbesserte WILDDESIGN-Expertise: CIDM
Stärkung der Marke über lokale Grenzen hinaus
Um das globale Ziel von REJOIN zu erreichen, hat WILDDESIGN eine Abteilung aus seinem eigenen Methodenbaukasten neu definiert und dabei geholfen, eine Designsprache zu entwickeln, die kohärente, hochfunktionale Produkte ermöglicht, die mit größeren, etablierten Unternehmen konkurrieren können. Diese neu optimierten Methoden führen letztlich zu einem kompakten Resultat: ein leicht verständliches, benutzerfreundliches Handbuch in dem das gesamte Produkt- und Benutzererfahrungs-Know-how sowohl als Leitfaden für zukünftige Entwicklungen als auch als unternehmensinterner Orientierungspunkt beschrieben ist. Wir nennen es das Corporate Industrial Design Manual oder kurz CIDM. Erfahren Sie mehr über unsere neue CIDM-Service-Seite ...
"Unser Produktdesign ist stark und die Designsprache einzigartig. Wir haben jetzt die Kernkompetenzen und das Vertrauen in sie erworben, um unseren schwierigen Markt mit großer Gewandtheit zu meistern."
Mr. Xu, CEO
Designsprache
Das CIDM, das wir für Rejoin entwickelt haben, beginnt mit der Einführung der Markenattribute und Werte, die dann zu den ergonomischen Voraussetzungen und zu einer Industrial Design Philosophie führen. Diese Kombination aus Funktion und Form wird dann im gesamten Handbuch angewendet und katalogisiert. Dabei werden Kapitel wie Form, Farbe, Logo-Platzierung, Material-Finish und sogar spezifische Herstellungsverfahren beschrieben, um das gewünschte Designergebnis zu erzielen. Methodisch stellen wir sicher, dass die nächste Entwicklung sowohl mit der Marke als auch mit den funktionalen Aspekten im Einklang steht, ohne dabei Kosten- und Zeitfaktoren zu negieren.
Das CIDM ermöglichte es REJOIN, eine beeindruckende Designpräsenz gegenüber dem großen globalen Wettbewerb zu schaffen. Eine Synergie zwischen Marken- und Produktidentität, die während des gesamten Produktentwicklungsprozesses angewendet werden kann, wodurch die Effizienz innerhalb der einzelnen Designphasen erhöht und die Kohärenz der Marke im Markt erhöht wird. Um mehr über die Vorteile von CIDMs zu erfahren, lesen Sie Teil 3 von „ Die Zukunft ist Nicht-invasiv“.
Nächste Schritte: Starkes designsprachliches Fundament
Marken- und Marketingsupport
Beginnend mit der Designkonzeption bis zu Rejoin’s Erfolg in China während der letzten 3 Jahre, verlagerte WILDDESIGN die professionelle Unterstützung immer weiter in Richtung Branding, Marketing und Verkauf. Während die erste Serie von Instrumenten grade klinisch validiert wird, wurde WILDDESIGN bereits mit der nächsten wichtigen Aufgabe betraut: die neuen Instrumente in einer Flaggschiff-Produktbroschüre zu visualisieren und zu präsentieren. Diese soll bei Besprechungen mit Krankenhauspersonal und Chirurgen verwendet werden. Die Broschüre ist der Beginn der öffentlichen Präsenz von Rejoin als neue ehrgeizige Sportmedizinmarke und auch wenn wir erst am Anfang dieses nächsten Kapitels stehen, wird sich das Rejoin-Erscheinungsbild weiter verfeinern, sobald wir Markt- und Kundenfeedback verstanden und implementiert haben.
Rejoin erkannte früh die Notwendigkeit, die Vielfalt aller gedruckten und digitalen Kommunikation aus einer Hand zu bekommen. Und so durfte WILDDESIGN weitere grafische und markenbezogene Unterstützung leisten, in Form von Standdesigns, Verpackungen und firmeninternen Unterlagen.


"Seit vielen Jahren verbindet uns die intensive Zusammenarbeit zwischen Rejoin und unserem Shanghai Team. Eine klare und unverwechselbare Design- und Brand- Direction hat uns von Anfang an zu diesem ausdrucksstarken Portfolio geleitet. Und jedes Jahr fügen wir ein paar weitere Instrumente hinzu..."
Ihr Ansprechpartner zum Projekt
Quan Shan
Head of Office Shanghai
+86 (0)21 5228 8112
quan.shan@wilddesign.cn




.webp)